But while researchers first predicted the existence of metallic hydrogen 80 years ago, the element has stubbornly refused to appear, even after we've raised pressures well above where it was expected to appear. Here, theorists have predicted, the electrons of hydrogen could be set free, converting the gas into a solid or liquid metal. Until these results are repeated, either by the Harvard tea-or preferably someone else-metallic hydrogen remains an exciting but unconfirmed possibility.Further Reading Strange, unpredictable chemistry at high pressureĬlose to the cores of gas giants, elements are squeezed by unimaginable pressures, capable of rearranging electron orbitals and playing havoc with the chemical bonding we see on Earth. Even once the sample is retested, there's room for doubt. Without some kind of confirmation of the experiment there's no way to know if this was some kind of fluke, no matter how rigorous the results might otherwise seem. Of all of these concerns, the lack of a second measurement is the most damning. "Now that the paper has been accepted, we're going to do further experiments." To preserve the material, he and Dias have kept it in the cryostat the lab has only two cryostats, and the other is in use for other experiments, he says. "We wanted to publish this breakthrough event on this sample," says. More tests are planned, but the researchers wanted to release what data they had before doing anything that might destroy the sample. It could also be the aluminum oxide that coats the diamonds. And the team never established that it is the hydrogen itself that is metallic. 
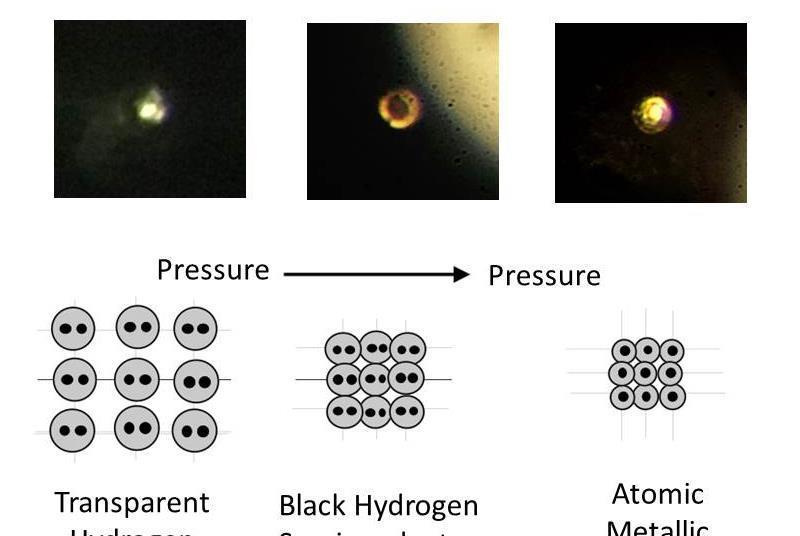
They never tested to see if it conducts electricity or had any other hallmarks of metallicity. Others are concerned that the team didn't repeat its experiment before publishing.įurther, the only evidence the team has that the hydrogen is metallic is its shiny appearance. Many other physicists are skeptical that the Harvard team managed to reach a pressure 20 percent higher than anyone else in the world on their first try. Right: The Harvard team claims the shiny sample at extreme pressures proves the existence of metallic hydrogen. Middle: Hydrogen at high pressure becomes dark. Left: Hydrogen at normal pressure is clear. This, the team says, is a clear sign the hydrogen has turned metallic. The hydrogen, which was initially clear, turned dark before becoming shiny at around 72 million psi. The team subjected a tiny drop of liquid hydrogen to intense pressures from their diamond anvil. Their research was published in the journal Science. But at that pressure, even the ultrahard diamonds can still shatter if they're not perfectly smooth and virtually flawless.īut one team from Harvard claims they've overcome this hurdle thanks to a new polishing technique, and have used their apparatus to produce real metallic hydrogen. Pressures at this scale, nearly 72 million pounds per square inch, can only be achieved with a powerful diamond anvil. However, creating Jupiter-level pressures in the laboratory is extremely difficult, which is why nobody has personally observed metallic hydrogen before. The electrical properties of theoretically metallic hydrogen could explain Jupiter's extremely powerful magnetic fields. Some scientists believe that metallic hydrogen exists in the center of Jupiter due to the extremely high pressures there. While the existence of metallic hydrogen has never been proven, there may be some evidence of it. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
